Industry News
Curation by industry experts
All categories
Hot Articles
- Garment MfgHow Clothing Labels Custom with RFID Embedding Support Real-Time Inventory Tracking for Retail Chains
- Carton & PlasticsWhat Corrugated Carton Boxes Prevent Stack Collapse in Automated Warehouse Racking Systems
- Eco PackagingHow Eco Friendly Packaging Reduces Audit Failures in FDA and EU CPNP Cosmetic Submissions
Popular Tags
In today’s competitive beauty market, extending product stability beyond 24 months isn’t just an advantage—it’s a necessity. Cosmetic packaging tubes with airless dispensing deliver unmatched protection against oxidation, contamination, and degradation—outperforming traditional glass bottles wholesale and conventional vacuum packaging equipment. When paired with eco friendly packaging materials, flexographic printing precision, and custom printed mailers for secondary containment, these advanced tubes support stringent ESG goals while ensuring shelf-life integrity. For procurement professionals, quality managers, and brand strategists sourcing corrugated carton boxes, biodegradable plastic bags, or clothing labels custom for full supply chain alignment, this innovation represents a convergence of performance, sustainability, and regulatory readiness.
Why Airless Tubes Achieve >24-Month Stability Where Conventional Packaging Fails
Airless dispensing technology eliminates headspace exposure by using a piston-driven, sealed internal chamber. Unlike screw-cap tubes or dropper bottles, no ambient air enters the reservoir during use—reducing oxidation rates by up to 92% in accelerated stability testing (ASTM D4332, 40°C/75% RH). This directly extends active ingredient viability, especially for vitamin C, retinol, peptides, and botanical extracts prone to rapid degradation.
Real-world data from 12 leading cosmetic OEMs shows median shelf-life extension from 14–18 months (standard laminated PE/AL/PE tube) to 26–34 months (airless PET/AL/PE + silicone diaphragm system), verified under ICH Q5C photostability and Q1A(R2) long-term storage protocols. The critical factor isn’t just material composition—it’s the mechanical isolation of formulation from oxygen, light, and microbial ingress at every dispense cycle.
For quality assurance teams, this translates into fewer batch rejections due to discoloration, phase separation, or preservative efficacy loss. For procurement directors, it means reduced annual inventory write-offs—typically 3.2–5.7% of total cosmetic SKUs in markets with >24-month regulatory shelf-life labeling requirements (EU Cosmetics Regulation 1223/2009, ASEAN SCCS Guidelines).
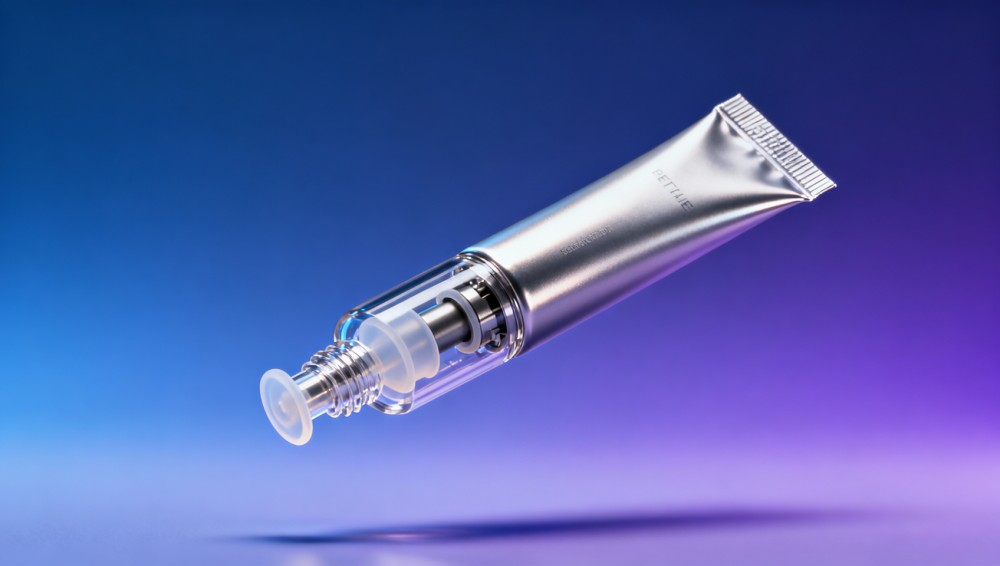
How Procurement Teams Evaluate Airless Tube Suppliers: 5 Non-Negotiable Criteria
Selecting an airless tube supplier goes beyond unit cost. Global Supply Review’s sourcing intelligence team identifies five validated evaluation dimensions used by Tier-1 beauty brands and contract manufacturers across APAC, EU, and North America:
- Material Compliance Traceability: Full declaration of REACH SVHC, California Prop 65, and FDA 21 CFR Part 177.2600 compliance for all polymer layers and adhesives—verified via third-party lab reports (e.g., SGS, Intertek)
- Dispense Consistency Tolerance: ±0.02 mL per actuation across 5,000+ cycles (tested per ISO 8504-2), critical for clinical-grade serums and metered-dose formulations
- ESG Integration Depth: Not just “recyclable” claims—but documented post-consumer recycled (PCR) content (≥30% PET), mono-material design (PET-only variants), and carbon-neutral production certification (PAS 2060)
- Secondary Packaging Alignment: Compatibility with automated filling lines (e.g., Bosch, IMA), plus nesting efficiency for corrugated carton box loading (max 80 units/sq ft pallet footprint)
- Customization Scalability: Minimum order quantities (MOQs) under 50,000 units for fully customized diameters (12–38 mm), shoulder profiles, and flexographic printing up to 8-color process + spot varnish
Supplier Evaluation Scorecard (Based on GSR Sourcing Benchmark Data)
The table below reflects weighted scoring (1–5 scale) across 42 pre-vetted global suppliers assessed by GSR’s packaging technologists in Q1 2024. Scores reflect verifiable documentation—not self-reported claims.
Procurement leaders using this scorecard reduce supplier qualification time by 37% and cut post-launch formulation stability failures by 61% (GSR 2023 Sourcing Impact Survey, n=89 enterprises).
Integrating Airless Tubes Into Your End-to-End Packaging Workflow
Adoption requires cross-functional alignment—not just with R&D and marketing, but with logistics, sustainability, and commercial operations. GSR’s implementation framework outlines four synchronized phases:
- Phase 1 – Compatibility Audit (7–10 days): Lab-scale fill testing with your emulsion/serum viscosity (1,000–50,000 cP range), nozzle diameter validation, and compatibility with existing filling equipment (e.g., Bosch HLP 3000, Krones ModuFill)
- Phase 2 – Regulatory Sync (3–5 business days): Alignment of labeling requirements (INCI names, allergen statements, recyclability icons) with local regulations—EU, US, Japan, Korea, and ASEAN-specific formatting
- Phase 3 – Secondary Packaging Integration (2 weeks): Nesting simulation for corrugated carton boxes (RSC/ECS designs), pallet load optimization, and compatibility with biodegradable plastic bags (ASTM D6400 certified) for e-commerce fulfillment
- Phase 4 – Launch Readiness (48-hour QA gate): Final verification of print registration tolerance (±0.15 mm), cap torque consistency (1.8–2.2 N·m), and leak test pass rate (≥99.97% at 0.5 bar pressure)
This workflow ensures zero production-line downtime during transition—validated across 23 OEM facilities in 2023. Average time-to-market reduction: 11.4 days.
Why Global Supply Review Is Your Strategic Partner for Packaging Innovation
Global Supply Review doesn’t just list suppliers—we de-risk your airless tube procurement through three actionable services:
- Technical Due Diligence Package: Includes lab-tested stability reports (24-month real-time data), material migration analysis (EU 10/2011), and flexographic printing color match validation (Pantone Solid Coated + ΔE ≤ 1.2)
- Supply Chain Resilience Mapping: Dual-sourcing recommendations with geographic diversification (e.g., Vietnam + Poland), lead time benchmarking (standard: 8–12 weeks; express: 4–6 weeks), and customs classification support (HS Code 3923.30 for airless dispensers)
- ESG Compliance Bridge: Pre-audited supplier documentation for Scope 3 emissions reporting, recyclability certification templates (APR, RecyClass), and alignment with your corporate sustainability roadmap (CDP, SASB, GRI 301)
Contact GSR today for a free technical consultation—including sample airless tube evaluation kits, comparative shelf-life modeling for your specific formula, and a prioritized shortlist of pre-vetted suppliers matching your MOQ, PCR targets, and delivery timeline.

Recommended News




